The Food and Drug Administration (FDA) has issued a public warning about the presence of counterfeit versions of several widely used over-the-counter (OTC) drugs. These fake OTC drug products pose a significant threat to consumer health and safety, and the FDA is urging the public to be vigilant when purchasing medications.
The affected OTC drugs include:
- Ibuprofen (Medicol® Advance) 200 mg Softgel Capsule
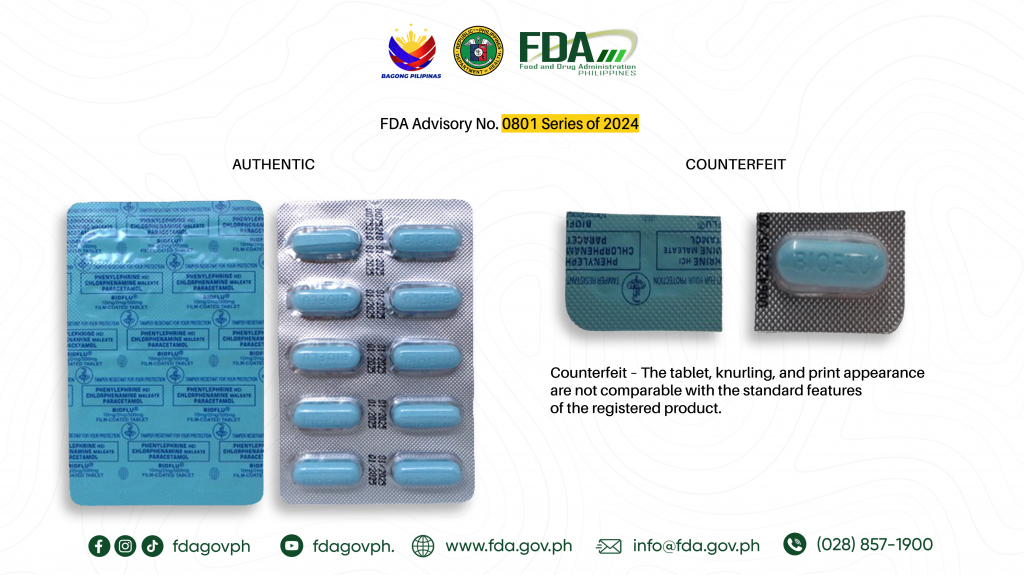
- Phenylephrine Hydrochloride + Chlorphenamine Maleate + Paracetamol (Bioflu®) 10mg/ 2mg/ 500mg Tablet
- Dextromethorphan Hydrobromide + Phenylpropanolamine Hydrochloride + Paracetamol (Tuseran® Forte) (Reformulated) 15 mg/25mg/325mg Capsule
- Aluminum Hydroxide + Magnesium Hydroxide + Simeticone (Kremil-S®) 178 mg/233 mg/30 mg Chewable Tablet
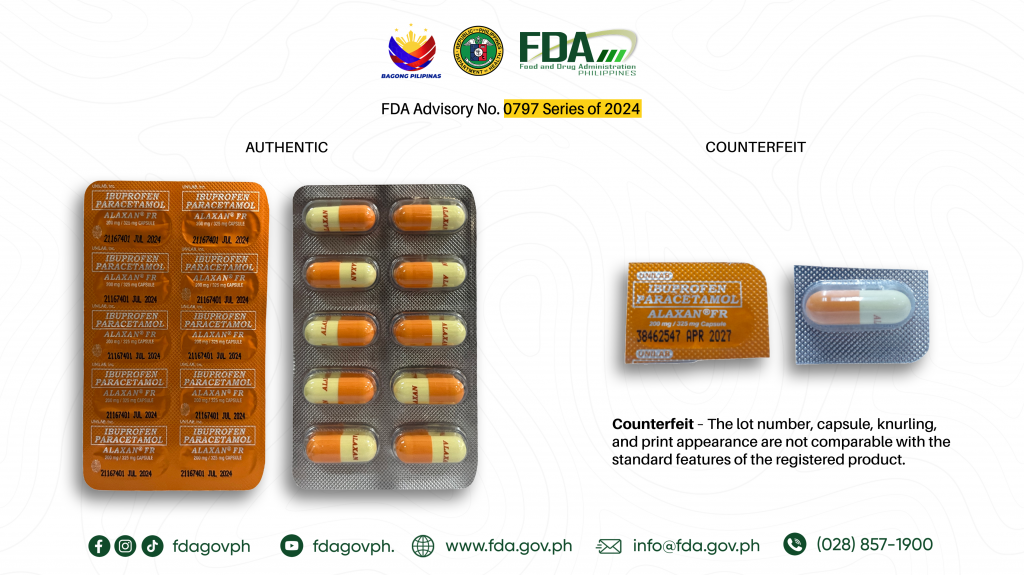
- Ibuprofen + Paracetamol (Alaxan® FR) 200mg/ 325mg Capsule
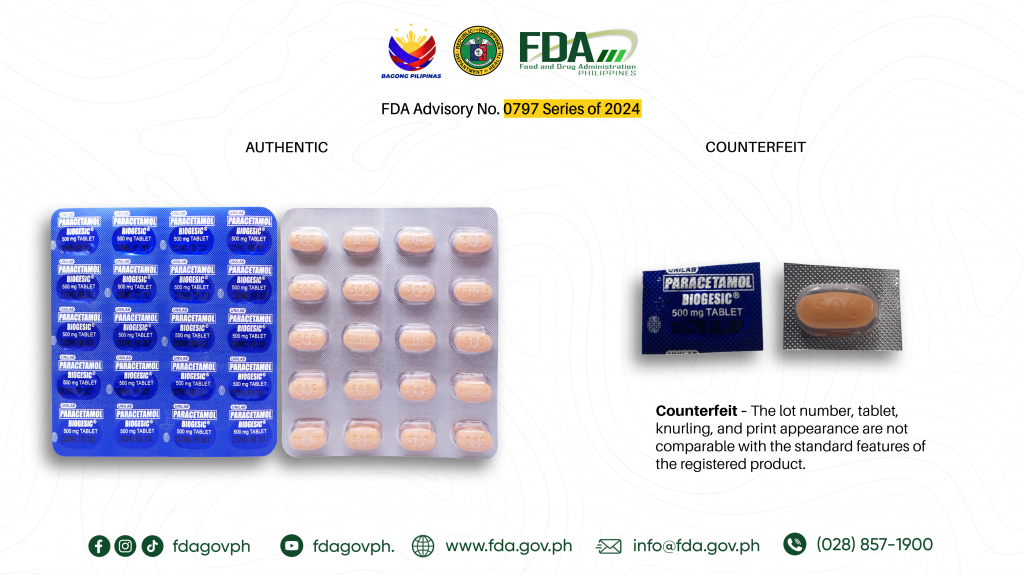
- Paracetamol (Biogesic®) 500mg Tablet
The Dangers of Counterfeit OTC Drugs
Counterfeit drugs are a growing global problem, and they pose a serious threat to public health. These fake medications may contain harmful substances, incorrect dosages, or no active ingredients at all. Consuming counterfeit drugs can lead to severe health complications, including organ damage, allergic reactions, and even death.
Protecting Yourself from Counterfeit OTC Drugs
To safeguard your health, it is crucial to purchase drug products only from FDA-licensed establishments. Avoid buying medications from unauthorized sources, such as online pharmacies or street vendors, as these are more likely to sell counterfeit products. If you suspect that you have purchased or consumed a counterfeit drug, discontinue use immediately and consult with a healthcare professional. Be sure to report any adverse reactions or suspected counterfeit products to the FDA through their online reporting facility, eReport, or by contacting the Center for Drug Regulation and Research.
The Role of Healthcare Professionals and Authorities
Healthcare professionals play a vital role in identifying and reporting counterfeit drugs. They should be vigilant in recognizing suspicious packaging, labeling, or product characteristics, and report any concerns to the FDA. Local Government Units (LGUs) and Law Enforcement Agencies (LEAs) are also responsible for ensuring that counterfeit drugs are not sold or made available in their jurisdictions. By working together, healthcare professionals, authorities, and the public can help combat the spread of counterfeit drugs and protect the health of individuals.
Serious Issue
The presence of counterfeit drugs in the market is a serious issue that requires immediate attention. By being aware of the dangers of these products and taking proactive measures to purchase medications from reliable sources, individuals can protect themselves and their loved ones from potential harm. Remember, that it is worth the extra effort to ensure that the medications you take are safe and effective. Stay informed, be vigilant, and always prioritize your well-being when it comes to healthcare decisions.
FDA Advisory link FDA Advisory No.2024-0801 (515 kB) and FDA Advisory No.2024-0797-20240229150046 (983 kB)